Respiratory Syncytial Virus RSV Antigen Test
Rapid Check CLIA Waived, 20/Box
NEW Test for patients Under 6 and Over 60 for age!
A rapid, single-use in vitro diagnostic device that provides a qualitative result to aid in the diagnosis of respiratory syncytial virus viral infection.
- 510(k) Cleared and CLIA Waived for Prescription Use
- Detection of fusion protein of respiratory syncytial virus
- Nasopharyngeal swab and nasal aspirate specimen
- Specimens collected from symptomatic patients aged less than 6 and over 60 years of age
- Sold by the Box of 20 Test Kits
Features & Benefits:
- Rapid Detection & Patient Management - RSV infection results in 15 minutes!
- Flexible Specimen Types -
- Nasophyaryngeal swab and/or nasal aspirates
- Nasal aspirate less invasive - Premier Performance* -
- Positive Percent Agreement: 93.9%
- Negative Percent Agreement: 97.7%
*Nasopharyngeal swab and nasal aspirate performance. Refer to Instructions for Use for additional performance claims - CLIA Waived -
- Demonstrates minimal training required
- Capable of testing in point-of-care settings
- Aids in Medicare/Medicaid reimbursement - Flexible Sample Transport -
- Fresh, refrigerated or frozen - immediate testing not required
- Compatible with various viral transport medium
- Remote collection of specimen if necessary - External Controls Included -
- Color coded (Red/+, Blue/-) screw cap vials
- Faciliate routine quality control inspection - 24 Month Shelf Life - Shelf life that spans viral seasonality
Disease Profile
Pathogen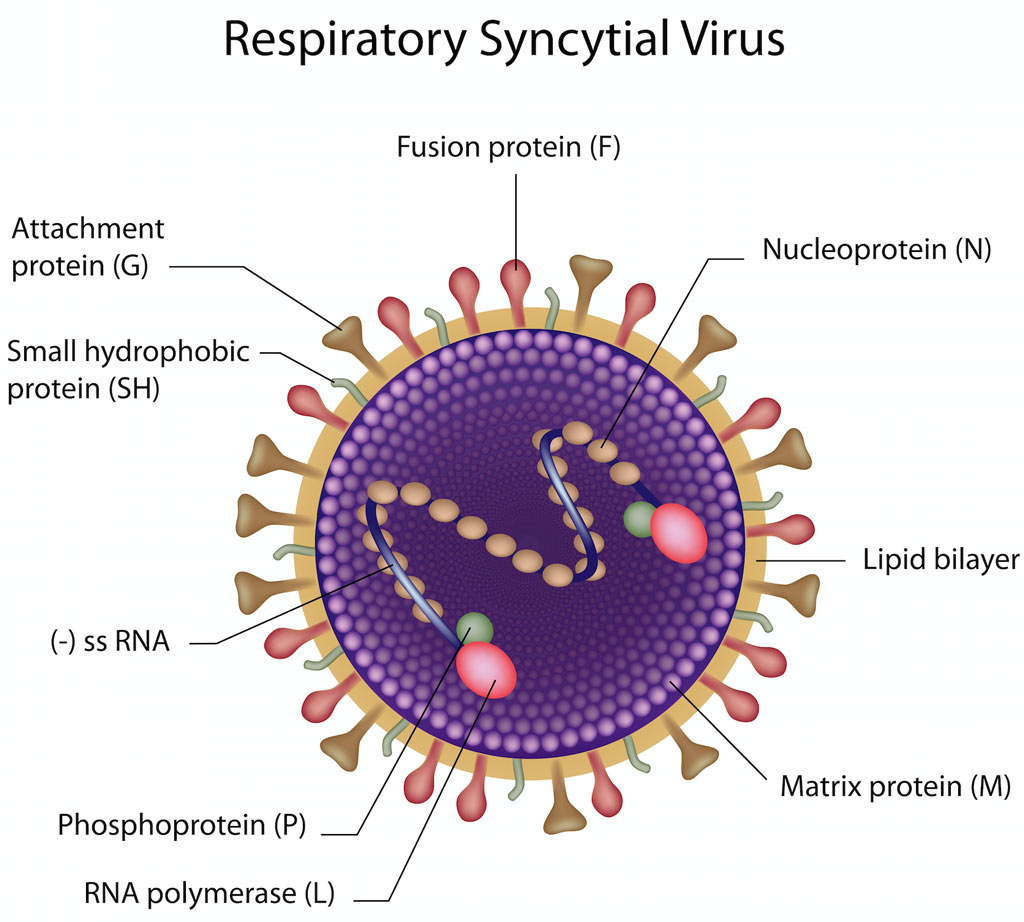
- Respiratory Syncytial Virus (RSV), Human orthopneumovirus
- Negative sense (-), single strand RNA
- Non-segmented genome – unlike influenza, RSV cannot differentiate into various subtypes
- Two subtypes: A and B
Transmission
- Infection via contaminated droplets circulated by coughing or sneezing
- Can survive on surfaces for 25 minutes
- Incubation period of 2 to 8 days
- Infected individual can remain contagious for 3 to 8 days
Symptomology
- Nasal congestion, Runny nose, Cough, Low-grade fever, Wheezing
- Due to similarities in symptomology, children between 1 to 5 years old may need differential test for respiratory illnesses
- RSV infects the respiratory tract. If left untreated 15-50% will develop a serious lower respiratory tract infection such as bronchiolitis, viral pneumonia, or croup
Seasonal Trends
- RSV season onset: mid-September to mid-November
- RSV season peak: late December to mid-February
- RSV season offset: mid-April to mid-May
- Patterns of circulation have been disrupted since the start of the COVID-19 pandemic (2020). RSV in southern regions begins to rise in spring and peaks in July
Epimeniology
Globally, RSV was estimated to have caused 66,000 to 199,000 pneumonia deaths in children less than 5 years old in 2005, making RSV the third most important cause of deadly childhood pneumonia after Streptococcus pneumonia and Haemophilus influenza[1]
High Risk Populations[1]
- Infants and young children
- Older adults, especially those 65 years and older
Epidemiology (United States)
- Most children under the age of 2 will be infected with RSV at least once.[2]
-
Leading cause of lower respiratory tract illness in infants (0-1 Years)
- Infant hospitalization rate, 0-1 years: 2,345 per 100,000 (151 for Influenza viruses)
- Total hospitalization rate, all age groups: 55 per 100,000 (64 for Influenza viruses)
-
1 in 13 children under the age of five required medical attention for RSV each year
- 60% of office visits were for 2 -5 years old
CDC Burden Estimates[3]
- Each year in the United States, RSV leads to approximately:
- 2.1M outpatient (non-hospitalization) visits among children <5 years old.
- 58,000-80,000 hospitalizations among children <5 years old.
- 60,000-160,000 hospitalizations among adults 65 years and older.
- 6,000-10,000 deaths among adults 65 years and older.
- 100–300 deaths in children younger than 5 years old.